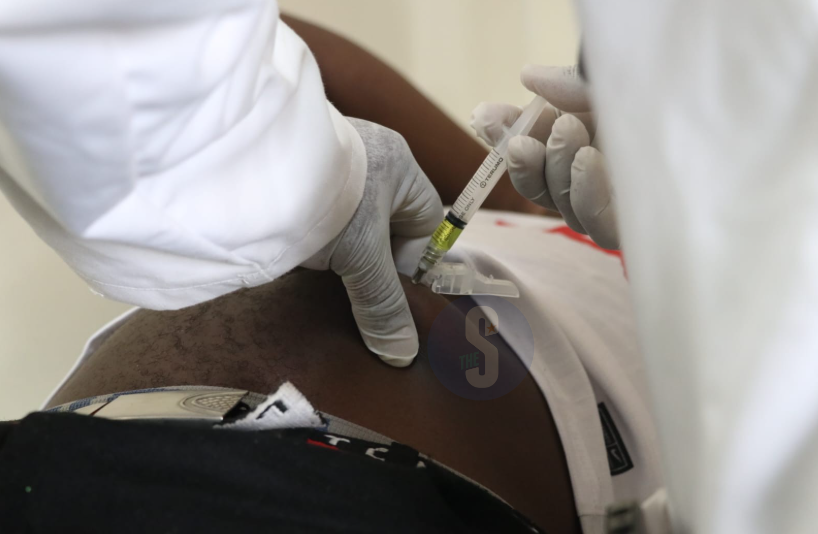
 Samson Mutua, the first patient to receive the Lenacapavir vaccine during the launch of the long-acting Prep at Riruta health centre, Nairobi on February 26, 2026. /LEAH MUKANGAI
Samson Mutua, the first patient to receive the Lenacapavir vaccine during the launch of the long-acting Prep at Riruta health centre, Nairobi on February 26, 2026. /LEAH MUKANGAIKenya on Thursday rolled out 21,000 vials of Lenacapavir in the first phase of administration of the anti-HIV injection targeting 15 high-risk counties.
Under guidelines issued by the World Health Organization, rigorous HIV testing is mandatory before initiating Lenacapavir to prevent the development of drug resistance, as the medicine acts as a potent antiretroviral.
Eligible recipients of the long-acting injectable HIV pre-exposure prophylaxis (Prep) must therefore undergo an HIV test and must not have been exposed to the virus within 72 hours prior to receiving the jab.
As part of its mandate to provide scientific guidance on Lenacapavir use, WHO conducted research to determine whether rapid diagnostic tests or self-tests should be used for the initiation or continuation of injectable Prep.
The systematic review compared evidence on different testing approaches for injectable Prep focusing on rapid diagnostic tests (RDTs), HIV self-tests (HIVST) and nucleic acid testing (NAT).
“We systematically searched for studies through October 18, 2024 that assess the use of different HIV testing strategies in the context of injectable long-acting Prep delivery. We also contacted authors and researchers from registered studies, identified through a survey, through December 1, 2024,” WHO said in its report.
The review selected 53 reports from 7,698 records across 15 countries in Africa, Asia, Europe and North and South America.
“Key findings from the review were that, when compared with HIV testing algorithms using lab-based testing and/or NAT techniques, RDT-based algorithms are likely to be more acceptable, feasible and affordable, may have faster turnaround time and could enable clients to link to ART after a positive diagnosis,” WHO said.
Seven studies reported no clinical or social harms associated with rapid diagnostic tests compared with lab-based testing or nucleic acid testing techniques.
The average turnaround time for results was same-day for RDTs, compared to a median of 35 days or more for lab-based testing.
WHO said HIV self-testing could increase testing frequency and enhance flexibility in service delivery, allowing users to test at any time.
Evidence also pointed to potential cost savings and high acceptability, feasibility and opportunities to diversify and streamline Prep implementation through demedicalised services such as self-care, pharmacy-based, telehealth and community-led approaches.
In conclusion, WHO said HIV testing strategies and algorithms relying on RDTs largely result in similar or better outcomes for users of injectable long-acting Prep compared with lab-based testing and nucleic acid testing techniques.
It said rapid diagnostic tests and HIV self-tests are highly acceptable to both users and healthcare providers due to their convenience, affordability and discreet nature, making them preferable to more invasive and time-consuming nucleic acid testing procedures.
The organisation added that rapid diagnostic testing could improve access to injectable Prep in resource-limited settings, particularly among marginalised populations and those in remote areas where nucleic acid testing resources are often unavailable.
“Implementing RDT-based testing is feasible due to their minimal infrastructure requirements, ease of use and immediate results, which facilitate ongoing Prep. This feasibility is particularly beneficial in limited resource settings and for mobile or underserved populations,” WHO said.
Kenya expects to receive an additional 12,000 continuation doses by April to support individuals initiated on the product.
Lenacapavir costs an estimated Sh7,800 per patient annually, but the Ministry of Health has clarified that the twice-yearly, long-acting injectable will be provided free of charge at designated public health facilities in 15 high-burden counties.
Two additional phases — one targeting 15 more counties and another focusing on 17 counties — will be rolled out later to progressively expand nationwide coverage as Kenya accelerates efforts to eliminate new HIV infections by 2030.
Lenacapavir is administered by a healthcare professional as a 1.5ml subcutaneous injection in the abdomen every six months.
The initial injection is accompanied by a “loading dose” of two 300mg tablets to enable the jab to reach therapeutic levels. Another two 300mg tablets are taken on day two.
The dosage is repeated six months from the date of the first injection, with a two-week window before or after.
Protection from HIV becomes effective on day three, two days after the injection and the oral loading tablets are taken.
Lenacapavir remains in the body at low levels for more than 12 months after the last injection.
This period, known as the “tail phase”, is characterised by insufficient drug levels to provide protection from HIV, and there is a risk of developing resistance if exposure occurs. Additional protection methods are recommended during this time.
Lenacapavir may be used during pregnancy. However, if pregnancy is planned or suspected, patients are advised to consult a healthcare provider for appropriate guidance and follow-up.
Patients should also inform their provider of all medications they are taking, as some drugs — including those used to treat tuberculosis — can reduce Lenacapavir levels and affect its effectiveness.
The functionality of prescriptions for erectile dysfunction may also be affected, potentially resulting in toxicity.
The most common side effects are injection site reactions such as nodules, pain, erythema, swelling and induration. Other side effects include headache and nausea.
Clinical trials have shown Lenacapavir to be 96 per cent effective in preventing HIV acquisition when administered correctly.







Comments 0
Sign in to join the conversation
Sign In Create AccountNo comments yet. Be the first to share your thoughts!